Revista Iberoamericana de Neuropsicología
Vol. 2, No. 2: 107-118, julio-diciembre 2019.
Recovery of cognitive, academic and adaptive functioning in pediatric anti-NMDAR: Serial neuropsychological data for a 16-year-old bilingual female
Mairim Vega, Psy.D1, Eyal Muscal M.D2, Karen Evankovich Ph.D2
1 Carlos Albizu University, Mayagüez Campus
2 Texas Children’s Hospital/Baylor College of Medicine
Correspondence information:
2431 Blvd Luis A Ferre
EDIFICIO A. PORRATA PILA
SUITE 205
Ponce, PR 00717-2113
mairimvegapsyd@gmail.com
[su_row][su_column size=”1/2″][su_button url=”https://neuropsychologylearning.com/wp-content/uploads/pdf/pdf-revista-vol2/vol2-n2-4-recovery-cognitive-academic-adaptive-functioning-pediatric-anti-nmdar.pdf” target=”blank” style=”flat” background=”#F38546″ color=”#ffffff” size=”6″ radius=”0″ icon=”icon: arrow-down” icon_color=”#ffffff” text_shadow=”0px 0px 0px #ffffff”]ARTÍCULO[/su_button][/su_column] [su_column size=”1/2″][su_button url=”https://neuropsychologylearning.com/wp-content/uploads/pdf/pdf-revista-vol2/vol2-n2-revista-iber-neuro-portada.pdf” target=”blank” style=”flat” background=”#F38546″ color=”#ffffff” size=”6″ radius=”0″ icon=”icon: arrow-down” icon_color=”#ffffff” text_shadow=”0px 0px 0px #ffffff”]REVISTA VOL2-2[/su_button][/su_column] [/su_row]
Recovery of cognitive, academic and adaptive functioning in pediatric anti-NMDAR: Serial neuropsychological data for a 16-year-old bilingual female
Objective: Anti-N-methyl-D-aspartate receptor encephalitis (anti-NMDAR), the second-most frequent cause of autoimmune encephalitis in children, has been associated with severe cognitive deficits disproportionate to the severity of the clinical presentation. Studies have suggested the presence of long-term persistent deficits in cognitive functioning. In this case study we present the clinical course description and cognitive recovery of a previously high functioning 16-year-old, bilingual, Hispanic female.
Method: We present serial neuropsychological data of a previously high-functioning 16-year-old female.
Results: Neuropsychological evaluations conducted at 5 and 9 months post symptom onset revealed persistent global deficits in cognitive, academic, and adaptive functioning necessitating repeat rituximab dosing and ongoing treatment with mycophenolate. Most areas of functioning improved to the average range by the 22-month follow-up although serum antibodies remained elevated.
Conclusion: Neuropsychological data proved to be a better indicator of the patient’s recovery of “real-world” functioning than clinical observations or serum antibody titers, underscoring the importance of neuropsychological evaluations for these patients.
Anti-N-methyl-D-aspartate receptor encephalitis (anti-NMDAR) is an autoimmune, inflammatory syndrome associated with a multi-stage and multi-domain neurologic illness that may initially present with neuropsychiatric symptoms but often progresses to a severe syndrome characterized by encephalopathy, seizures and abnormal movements. It has been identified as the second-most frequent cause of autoimmune encephalitis in children after acute disseminated encephalomyelitis (ADEM), with a much higher incidence in females (1, 2, 3). NMDAR may be more prevalent in children and young adults than many forms of viral encephalitis(4). It may be associated with ovarian teratomas in children over 12, but occurs more commonly without tumors (non-paraneoplastic) in pediatric populations, unlike in adults (1, 5). Initial presentation varies with age of onset, with adults and adolescents typically presenting with neuropsychiatric symptoms (e.g. anxiety, paranoia, delusions, cognitive changes)( 2, 5) and children younger than 12 years and toddlers/infants more commonly presenting with neurologic symptoms such as abnormal movements (2,4,21) or behavioral changes such as irritability and language dysfunction (3,6).
Disease course usually progresses to include memory disturbance, dyskinesias, altered consciousness and autonomic nervous system dysfunction (in approximately 40% of preadolescents and 50% of adolescents). Speech and language dysfunction is common in children, with more than 80% presenting symptoms such as mutism and echolalia(6). Treatment may include surgical removal of the teratoma in paraneoplastic cases, first-line immunosuppression with high dose steroids, intravenous immunoglobulins (IVIG), and/or plasma exchange, and second-line immunosuppression with rituximab and/or cyclophosphamide if symptoms are not improving,(7, 8, 9). With immunotherapy and tumor removal, about 75-85% of patients show substantial improvement in their clinical presentation. Recovery time can be highly variable, with a minority of individuals recovering within several months and others recovering over years, with continued improvements documented up to 18-24 months following the onset of symptoms (7, 10, 11). Full recovery is less likely when there is no identified tumor, but in such cases, second line immunotherapy can often be effective. Patients receiving early treatment (within 4 weeks of symptom presentation) show significantly better outcomes than those treated later during the disease course (2,7), and longer hospitalization times being linked to residual cognitive deficits at follow-up on for some cohorts(20). Outcome data in the few existing observational cohort studies have relied on crude assessments of function (such as modified ranking scores) (1,7,21).
Although the clinical features of childhood onset anti-NMDAR have been better characterized in the last 5 years, there is still a paucity of information on long-term cognitive outcome. For adults, severe cognitive deficits disproportionate to the severity of the clinical presentation have been documented early in the disease process (12), and persistent deficits in attention, executive functions, and memory have been documented months to years following apparent clinical recovery(10,13,14). Similarly, some pediatric studies have suggested persistent deficits in executive functions, attention, memory, language, processing speed, self-care and adaptive functioning in general, although available neuropsychological data is sparse (2, 15, 16, 17, 22, 23). To our knowledge, data documenting academic functioning in pediatric anti-NMDA receptor encephalitis has yet to be described.
A description of a bilingual adolescent with CSF, serum antibody proven anti-NMDAR treated with standard of care interventions, who was followed closely with serial neuropsychological evaluations, will be provided. Her initial inpatient course and subsequent follow up transpired at the same medical center which allowed for documentation of her full recovery. IRB approval was obtained to abstract data on her clinical course, serologic studies and neuropsychological evaluations. Signed, written consent was obtained from the patient and family.
Serial neuropsychological testing was carried out using the following instruments, administered in English and using the normative data for U.S. based populations provided for each test by the respective test authors: Wechsler Adult Intelligence Scale, Fourth Edition; Expressive Vocabulary Test, second edition; Grooved Pegboard, The Beery-Buktenica Developmental Test of Visual-Motor Integration, sixth edition; Wide Range Assessment of Memory and Language, second edition (select subtests); Kaufman Test of Educational Achievement, second edition. The Test de Vocabulario en Imágenes Peabody, was also administered in Spanish, and scored using the Mexican normative sample. Finally, the patient’s adaptive functioning was assessed by administering the Vineland Adaptive Behavior Scales, second edition, Spanish Survey Interview to her mother and using the U.S. based normative data to score the instrument. Her premorbid cognitive functioning was estimated using school documentation of her academic performance and test results were compared across all three serial neuropsychological evaluations in order to determine cognitive deficits and recovery.
The patient was a previously healthy, 16-year-old, bilingual (Spanish-English speaking) Hispanic female living in the United States (U.S.), with no prior concerns regarding her functioning. The primary language spoken at home was Spanish, although the patient had been exposed to English since pre-school, and participated in English only classes in school since the second grade. She was enrolled in an advanced 10th grade curriculum in a public high school in the U.S. at the time of symptom onset and was considered an excellent student, with good social functioning and involvement in several honor societies and volunteer groups. Eleven days prior to hospital admission she requested assistance to change her clothes because she was unable to use her right hand. She was promptly evaluated by her primary care physician who attributed this symptom to stress. Three days later she was referred to a psychologist by school personnel, to whom she reported auditory hallucinations (hearing voices), insomnia, and extreme forgetfulness. She was observed to have paucity of speech, saying only “yes”, “no”, and “I don’t know” during the session. She was then sent to an emergency room, where MRI and blood tests were normal except for elevated thyroid stimulating hormone (TSH) and a urinary tract infection. Psychiatric admission was recommended, but no beds were available. The patient returned to her primary care physician who reportedly prescribed an unspecified “relaxant.” Ten days after symptom onset she presented with severe disorientation in school. She was confused and unable to locate her classes even when a printed schedule was provided. The school nurse contacted the patients’ primary care physician, who referred her to a tertiary pediatric hospital for a full evaluation due to severe cognitive impairment and concern for encephalopathy.
The patient was admitted to the hospital eleven days after symptom onset, where an extensive medical work-up revealed high titer NMDAR antibodies in both serum and CSF. MRI findings revealed no atrophy or lesions, and electroencephalogram (EEG) studies found continuous generalized slowing, more prominent in left frontal, left temporal and left temporo-central regions initially. In addition to psychiatric symptoms (auditory and visual hallucinations,), the patient demonstrated the following symptoms: right hemiparesis, waxing and waning mental status consisting of confusion and disorientation, sleep disturbance, memory problems, and aphasia characterized by reduced verbal expression and perseverative language. Symptoms later progressed to autonomic instability, dysphagia requiring nasogastric tube placement, choreiform movements, and amenorrhea.
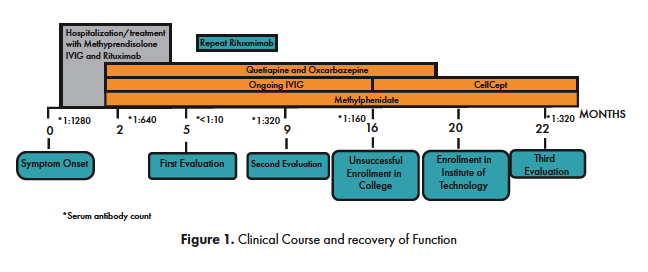
Starting 13 days after symptom onset the patient was treated with first line-immunosuppression per institutional best care practices. This included high dose pulse methylprednisolone (8 doses total of 1 gram each during the first 3 weeks), IVIG (2 g/kg split over a five-day course), as well as six cycles of plasma exchange due to limited response to initial medical management. Second line therapy of rituximab (500 mg/2 at 1 gram for 2 doses) was initiated to deplete autoantibody producing lymphocytes (B cells) due to lack of initial response and worsening encephalopathy. Clinical focal seizures consisting of twitching of the right hand and face, aphasia, and confusion, were associated with activity arising from left temporo-central region. These were initially treated with lorazepam and levetiracetam, and later with oxcarbazepine. Her psychiatric symptoms were treated with an atypical antipsychotic (quetiapine). She initially required propranolol for blood pressure control and management of dysautonomia but was later titrated off this medication without complication. She spent 6 weeks on the general neurology floor, requiring transfer to the PICU for management of worsening symptoms for 2 days out of her total length of stay. She was then transferred to the hospital’s inpatient rehabilitation unit (IRU), where she received physical, occupational and speech/language therapy for four weeks. In the IRU, her neurobehavioral status was monitored by neuropsychology, but significant, persistent altered mental status rendered her untestable. She demonstrated improvements in alertness, orientation, ability to follow commands, mobility and activities of daily living. She was discharged home 11 weeks after symptom onset under continued treatment with monthly IVIG, quetiapine and methylphenidate. [See Figure 1 for clinical course and treatment.]
Initial Neuropsychological Evaluation
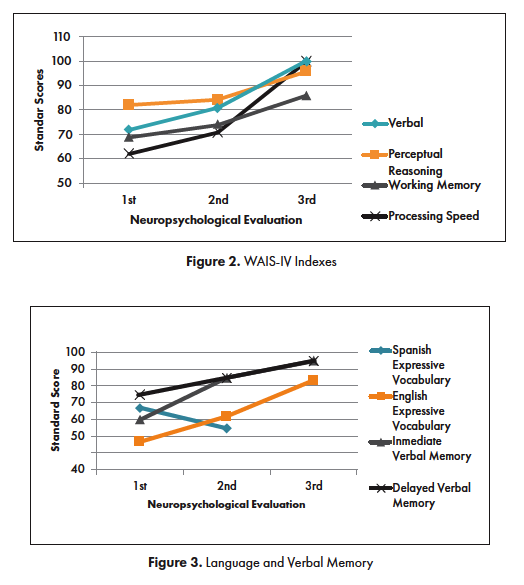
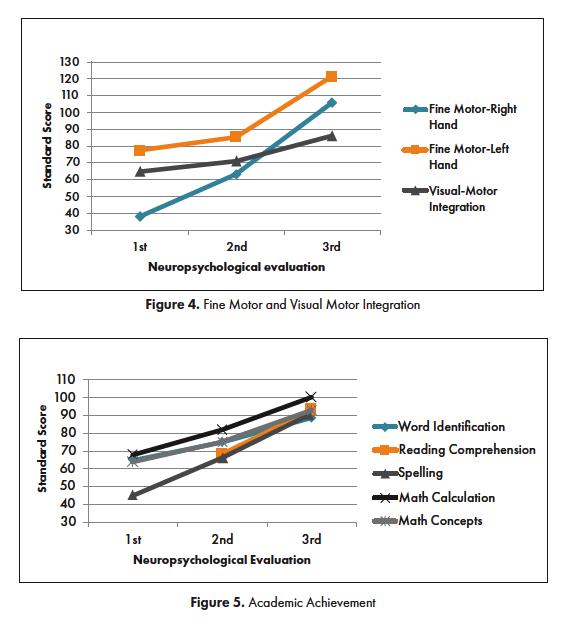
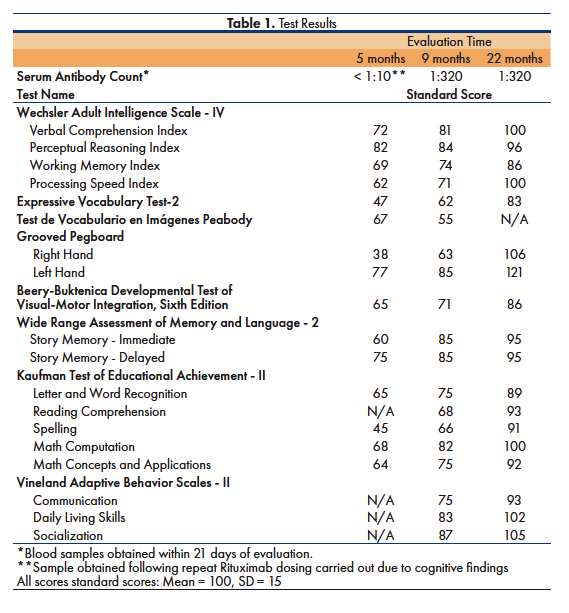
Approximately 5 months after initial symptoms the patient was receiving homebound school instruction but expressed a desire to return to school. Her clinical presentation during a follow-up with pediatric neurology suggested that she might be able to return to school, so she was referred for a comprehensive outpatient neuropsychological evaluation to identify accommodations that would facilitate her return to school. She took her prescribed quetiapine for the first testing session, and both methylphenidate and quetiapine for her second session. She demonstrated some awareness of her persistent cognitive and psychiatric symptoms and appeared social. Her presentation was remarkable for blunted affect, slow processing speed and significant word finding problems. She was unable to simultaneously eat a snack and participate in a conversation. The content of her spontaneous language was limited, vague, and perseverative, particularly on the subject of returning to school. She was noted to have more difficulties expressing facts in Spanish than in English, and could not correctly identify her year of birth in Spanish, although she did identify it correctly in English. The neuropsychological evaluation was carried out by a bilingual clinician and included both English and Spanish measures. The majority of the measures were administered in English given her apparent difficulties communicating facts in Spanish. However, Spanish instruments were administered in Spanish using standardized Spanish procedures. Her premorbid level of intellectual functioning was estimated to be at least average, based on her excellent performance within an advanced academic curriculum, as evidenced in school documents provided by her parents. The neuropsychological evaluation, however, revealed profound global impairments, most notably in processing speed, memory, expressive language, and fine-motor coordination with right hand more impaired than left hand. [See Figures 2-5 for test results]
Neurocognitive findings, coupled with the return of intermittent visual hallucinations and paranoid ideation, prompted the following adjustments in her treatment: increased IVIG schedule (from monthly to every three weeks), repeat rituximab (2 additional doses once it was apparent that B cells were depleted), added methylprednisolone pulse dosing (2x), and increased quetiapine dosage. The family reported a decrease in psychotic symptoms and an increase in social and academic functioning following these adjustments. Given the severity of her cognitive and academic deficits, homebound special education instruction was recommended to re-teach academic skills that had been lost and to allow time for further recovery prior to returning to campus. Speech/language therapy and occupational therapy were recommended to address her persistent language and motor deficits. Care was taken to provide the parents with information about NMDAR encephalitis and the recovery process in Spanish, their native language, and to support them in the process of obtaining services for the patient.
Second Neuropsychological Evaluation
The patient underwent a second neuropsychological evaluation approximately 9 months after symptom onset, having taken her prescribed methylphenidate and quetiapine. Notable improvements in affect, alertness and reciprocal conversation were observed. Although she was relatively faster and more flexible when responding to questions, her presentation continued to be remarkable for slow processing speed and word finding problems in English and Spanish. She required frequent repetition of instructions. Test results revealed persistent, global cognitive deficits. Slightly higher scores were obtained on measures of memory, expressive English vocabulary, verbal reasoning, processing speed, fine motor coordination (although marked right hand incoordination persisted), and academic skills [See Figures 2-5 for test results].
Given her estimated premorbid level of functioning (based on a documented excellent academic performance in an advanced school curriculum), some improvement was noted but she had not returned to the estimated baseline. Spanish receptive vocabulary did not appear to show improvement on standardized testing. Recommendations for school included ongoing special education supports and consideration of an additional year of school. She was again referred for outpatient speech/language therapy, which she received weekly for the next four months. She also received outpatient physical therapy (PT). Approximately 10½ months after symptom onset (April, 2015), she returned to school half-time to complete her senior year. She had significant difficulties adjusting to the classroom setting and keeping up with classes, but was allowed to graduate due to her pre-illness academic accomplishments.
Treatment with IVIG ended 13 months after symptom onset, but treatment continued with maintenance phase immunotherapy (mycophenolate), due to the continued presence of high titer serum antibodies and the protracted course of her initial disease presentation. Both oxcarbazepine and quetiapine were discontinued 16 months after symptom onset; treatment with methylphenidate was maintained.
The patient enrolled at a state college near her home but struggled to sustain her attention and keep up with her course work. After one month, she decided to postpone college due to her cognitive difficulties. She applied for several jobs but significant anxiety interfered with her performance during interviews. Nonetheless, continued gradual improvement was noted. By 19 months after symptom onset, she became fully independent with activities of daily living, driving, and managing her appointments and medications. She began a pharmacy technician course at an institute of technology. At the time of her third evaluation, she was obtaining all A’s in her classes. Treatment with methylphenidate and mycophenolate continued.
Third Neuropsychological Evaluation
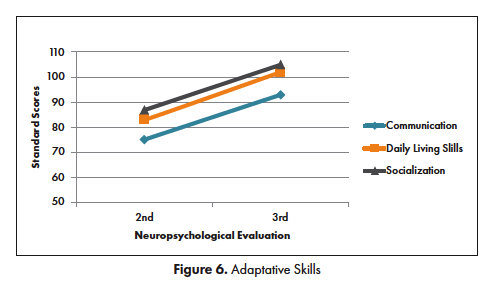
A follow-up evaluation was conducted 22 months after symptom onset. The patient took methylphenidate prior to the assessment. Serum antibodies remained elevated per blood tests completed at the time of the evaluation. Significant improvements were noted in alertness, attention, mental flexibility, and interpersonal engagement. Persistent word finding problems were noted but she more easily followed conversations and responded to questions. Objective test results showed significant, global improvements in cognitive functioning. Overall intelligence, memory, mental flexibility, and fine motor skills improved to the Average range. Persistent, subtle weaknesses were noted in working memory and processing speed, although considerable improvement was noted. Academic functioning improved to the Average range, with the exception of her performance on academic fluency measures, reflecting the subtle, persistent difficulties processing speed and sustained attention. Spanish receptive vocabulary could not be evaluated because the patient had aged out of the established age range for the test that we had available. Recommendations included school accommodations to reduce the impact of inattention and slower processing on academic performance and follow-up with the state department of rehabilitation. [See Figures 2-5 and Table 1 for test results]
The clinical course description and cognitive recovery of a previously high functioning 16-year-old, bilingual, Hispanic female was presented. To the knowledge of the authors, this is the first case study to document the recovery of impairments in academic functioning as a consequence of childhood-onset anti-NMDAR with serial, standardized assessments. The patient’s clinical course was similar to that of adults with anti-NMDAR, with a predominance of psychiatric symptoms identified early in the disease along with right-sided motor difficulties, which evolved to a right hemiparesis with disease progression. Initial symptoms also included speech dysfunction, which is commonly noted in preadolescent children (6). While improvement was noted, persistent right –sided fine motor weakness and word finding problems were evident in all evaluations, perhaps suggesting greater left hemisphere involvement. Consistent with previous literature, MRI findings were not significant, but EEG studies showed general slowing (3, 17, 18), at times in the left frontal, left temporal and left temporo-central regions.
Treatment with immunotherapy was initiated within 13 days of initial symptom presentation. Early intervention with immunotherapy has been correlated with more positive outcomes. As with many pediatric patients, both first- and second-line immunotherapies were necessary. Improvements were observed in clinical symptoms and social functioning 5 months after symptom identification, which lead her parents, physicians, and educators to conclude she was ready to return to school. Neuropsychological evaluations conducted 5 and 9 months after symptom onset, however, revealed global deficits in cognitive, academic, and adaptive functioning necessitating an Individual Education Plan with targeted therapies as well as conferences with school personnel to educate them about anti-NMDAR and her expected recovery. Attempts at continuing her education as she had premorbidly were unsuccessful, consistent with neuropsychological test findings.
Serial neuropsychological data obtained over 22 months documented the protracted recovery of cognitive deficits following anti-NMDAR encephalitis. Although gradual increases in test scores were noted during each evaluation, she did not achieve Average range levels of functioning until the 22-month follow-up. Academic skills were impaired initially and showed a similar pattern of recovery as cognitive functioning, highlighting a global impact of the disease on brain functioning. Previous research indicates that deficits in specific functions such as memory and executive functioning can persist months to years following symptom onset (10,13,14). In this patient, subtle weaknesses in working memory, processing speed, and word finding in English and Spanish were documented at 22 months after symptom onset.
Despite the recovery noted in her cognitive and academic skills, the patient’s serum antibodies were elevated at the time of her third follow-up. Serum antibodies were lower at the time of previous neuropsychological evaluations and she had continued treatment with mycophenolate at the time of the 22 month follow-up. Despite fluctuations in serum antibodies over the 22-month period, progressive recovery was noted in cognitive functioning with ongoing mycophenolate treatment. Recovery of cognitive functions was reflected in concomitant improvements in academic and adaptive functioning.
The few existing adult studies report a weak correlation between cognitive outcomes and antibody titers in cerebrospinal fluid (CSF) and serum at disease onset (13). Serum studies have been found to be even less specific than CSF antibody titers at monitoring disease. Lebon et.al. (15) described a 16-year-old girl whose rapid decrease in serum antibody titers with treatment was not associated with a rapid improvement in cognitive functioning making it difficult to associate cognitive improvement with changes in antibody titers. In the present case, serum antibody studies were obtained within two weeks of the initial evaluation, several times during her course of recovery, and at the time of the third evaluation. [See Figure 1 for details]
Greda-Arribas et al. (19) found that CSF titers were more sensitive than blood serum antibodies, and that changes in CSF titers were more closely related to clinical relapses. In this study, 13.2% of patients without detectable serum antibodies on cell-based arrays continued to have NMDA receptor antibodies present in CSF. The study also found that patients with high antibody titers and those demonstrating only small reductions in CSF antibodies during the first four months of disease were less likely to show good outcome. Most patients showed a decrease in serum and CSF titers, regardless of their cognitive outcome, suggesting that the results of a neuropsychological assessment may be more important for decision making (e.g. provision of immunomodulatory therapy, educational supports, or ancillary intervention such as occupational or speech therapies) than serum titers alone. Our patient showed undetectable serum antibodies at 23-week testing following repeat rituximab dosing, but serum antibodies were present at all other testing dates. CSF titers were only obtained during the initial work-up, as it is often not possible to obtain longitudinal CSF titers in pediatric patients and thus, a comparison could not be made.
Neuropsychological data proved to be a better indicator of the patient’s recovery of “real-world” functioning (e.g. ability to function in school, level of independence), than serum antibodies and clinical observation. This was true for the patient described by Lebon et.al. (15) who, despite apparent spontaneous improvement in mood and behavior, showed persistent memory and executive function deficits in neuropsychological evaluation conducted at 9-month follow-up. An adult case presented by Finke et.al.(13) also showed severe cognitive dysfunction on neuropsychological evaluation disproportionate to the severity of the clinical presentation.
Present findings underscore the importance of neuropsychological data for making clinical decisions and return-to-school recommendations for pediatric patients recovering from NMDAR encephalitis. They provide important information about cognitive, academic, and adaptive functioning. As documented in the current case, serial neuropsychological evaluations provided valuable information regarding recovery of function and aided in decision-making regarding treatment and recommendations. Given that the patient was still in the recovery process, re-evaluations of cognitive function were recommended at least once a year to modify recommendations as needed. Similarly, it is recommended that serial neuropsychological evaluations be routinely carried out with pediatric patients recovering from NMDAR encephalitis to monitor recovery and aid in clinical decisions and return-to-school recommendations. We believe that neuropsychology services should be an integral part of the care of children with anti-NMDAR and other similar autoimmune encephalitides (AE). Any center of excellence model for AE care should take into account the integration of these and other mental health services in a multi-disciplinary care team.
Case Limitations and Recommendations for Further Study
When evaluating multi-lingual and multi-cultural patients, it is important to take into account how these factors can impact test results. Our case study was limited by the lack of additional tests to evaluate Spanish language recovery in the patient, as compared to the recovery of English language skills. Further studies with bilingual and multilingual patients should aim to document and compare the recovery of all language functions. Additionally, further neuropsychological studies with larger cohorts of pediatric anti NMDAR patients are recommended to overcome the limitations inherent to case studies during the process of characterizing the relationship between antibodies titers and cognitive functioning, as well as to further characterize the course and long-term outcome of neurocognitive and academic impairments resulting from this disease in the pediatric population.
All authors confirm that there are no known conflicts of interest associated with this publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Acknowledgements
Maria Grosch.
St. Louis Children’s Hospital/ Department of Psychology
- Dalmau J, Lancaster E, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R. Clinical experience and laboratory investigations in patients with anti-NMDAR encephalitis. Lancet Neurol. 2011 Jan;10(1): 63-74: 63-74.
- Armangue T, Titulaer MJ, Málaga I, Bataller L, Gabilondo I, Graus F, et al. Pediatric anti-N-methyl-D-aspartate receptor encephalitis—clinical analysis and novel findings in a series of 20 patients. J Pediatr. 2013 Apr; 162(4): 850-856
- Brenton JN, Schwartz RH, Madoo L. Anti-NMDA-receptor encephalitis: A review. Contemporary Pediatrics. 2015 Jul 11; 32(7). Available from: http://contemporarypediatrics.modernmedicine.com/ contemporary-pediatrics/news/anti-nmda-receptor-encephalitis-review
- Gable MS, Sheriff H, Dalmau J, et al. The frequency of autoimmune N-methyl-D-aspartate receptor encephalitis surpasses that of individual viral etiologies in young individuals enrolled in the California Encephalitis Project. Clin Infect Dis. 2012 Apr; 54(7): 899-904.
- Peery HE, Day GS, Dunn S, Fritzler MJ, Pruss H, Doja A, et al. Anti-NMDA receptor encephalitis. The disorder, the diagnosis and the immunobiology. Autoimmun Rev. 2012 Mar 11;11: 863-872.
- Florance NR, Davis RL, Lam C, Szperka MD, Zhou L, Ahmad S, et al. Anti–N‐methyl‐D‐aspartate receptor (NMDAR) encephalitis in children and adolescents. Ann Neurol. 2009 May 28; 66(1):11-18.
- Titulaer MJ, McCracken L, Gabilondo I, Armangué T, Glaser C, Iizuka T, et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol. 2013 Jan 3; 12(2): 157-165
- Lazar-Molnar E, Tebo AE. Autoimmune NMDA receptor Encephalitis. Clin Chim Acta. 2015; 438: 90-97
- Dale RC, Brilot F, Duffy LV, Twilt M, Waldman AT, Narula S, et al. Utility and safety of rituximab in pediatric autoimmune and inflammatory CNS disease. Neurology. 2014 Jul 8; 83:142–50
- Wright S, Hacohen Y, Jacobson L, Agrawal S, Gupta R, Philip S, et al. N-methyl-D-aspartate receptor antibody-mediated neurological disease: Results of a UK-based surveillance study in children. Arch Dis Child. 2015 Jun 1;100(6): 521-526
- Byrne S, McCoy B, Lynch B, Webb D, King MD. Does early treatment improve outcomes in N‐methyl‐D‐aspartate receptor encephalitis? Dev Med Child Neurol. 2014 Mar 19; 56(8):794-796
- Vahter L, Kannel K, Sorro U, et al. Cognitive dysfunction during anti-NMDA-receptor encephalitis is present in early phase of the disease. Oxf Med Case Rep. 2014 Jul 1;4:74-76.
- Finke C, Kopp UA, Prüss H, Dalmau J, Wandinger KP, Ploner CJ, et al. Cognitive deficits following anti-NMDA receptor encephalitis. J Neurol Neurosurg Psychiatry. 2013 Apr; 83(2):195-198
- Kayser MS, Dalmau J. Anti-NMDA receptor encephalitis in psychiatry. Curr Psychiatry Rev. 2011 Aug 1;7(3): 189
- Lebon S, Mayor-Dubois C, Popea I, Poloni C, Selvadoray N, Gumy A, et al. Anti-N-methyl-D-aspartate (NMDA) receptor encephalitis mimicking a primary psychiatric disorder in an adolescent. J Child Neurol. 2012 Feb;27(12):1607-1610.
- Iadisernia E, Battaglia FM, Vanadia E, Trapolino E, Vincent A, Biancheri R, et al. Anti-N-Methyl-d-aspartate-receptor encephalitis: Cognitive profile in two children. Eur J Paediatr Neurol. 2012 Jan 1;16.1: 79-82.
- Houtrow AJ, Bhandal M, Pratini NR, Davidson L, Neufeld JA. The Rehabilitation of Children with Anti-NMDA-Receptor Encephalitis: A Case Series. Am J Phys Med Rehabil. 2012 May 1; 91(5): 435-441.
- Suthar R, Saini AG, Sankhyan N, et al. Childhood Anti-NMDA Receptor Encephalitis. Indian J Pediatr. 2016 Jul 1; 83(7): 628-633.
- Greda-Arribas N, Titulaer MJ, Torrents A, McCracken L, Leypoldt F, Gleichman AJ, et al. Antibody titres at diagnosis and during follow-up of anti-NMDA receptor encephalitis: A retrospective study. Lancet Neurol. 2014 Feb; 13(2):167-77.
- Nunneley C, Lotze T, Muscal E. Risk of long-term neuropsychiatric impairment in pediatric anti-NMDA receptor encephalitis (P3. 197). Neurology. 2017 Apr 18;88(16 Supplement):P3-197.
- Granata T, Matricardi S, Ragona F, Freri E, Zibordi F, Andreetta F, at al. Pediatric NMDAR encephalitis: A single center observation study with a closer look at movement disorders. European Journal of Paediatric Neurology. 2018 Mar 1;22(2):301-7.
- Gordon-Lipkin E, Yeshokumar AK, Saylor D, Arenivas A, Probasco JC. Comparative Outcomes in Children and Adults With Anti-n-methyl-d-aspartate (anti-nmda) Receptor Encephalitis. Journal of child neurology. 2017 Oct;32(11):930-5.
- De Bruijn MA, Aarsen FK, Van Oosterhout MP, Van Der Knoop MM, Catsman-Berrevoets CE, Schreurs MW, et al. Long-term neuropsychological outcome following pediatric anti-NMDAR encephalitis. Neurology. 2018 May 29;90(22):e1997-2005.