Revista Iberoamericana de Neuropsicología
Vol. 3, No. 1: 15-28, enero-junio 2020.
The impact of maternal prenatal complications on verbal fluency measures in children with ADHD: an empirical study
Paula Karina Perez, PsyD.1, Isaac Tourgeman, PhD.2
1 Rusk Rehabilitation at New York University Langone Health, New York, USA
2 Department of Psychology. Albizu University, Miami, USA
Corresponding author:
Paula Karina Perez, PsyD.
New York University Langone Health
Rusk Rehabilitation
Phone: +1 (305) 336-6950
E mail: paukaperez@gmail.com
[su_row][su_column size=”1/2″][su_button url=”https://neuropsychologylearning.com/wp-content/uploads/pdf/pdf-revista-vol3/vol3-n1-2.pdf” target=”blank” style=”flat” background=”#F38546″ color=”#ffffff” size=”6″ radius=”0″ icon=”icon: arrow-down” icon_color=”#ffffff” text_shadow=”0px 0px 0px #ffffff”]ARTÍCULO[/su_button][/su_column] [su_column size=”1/2″][su_button url=”https://neuropsychologylearning.com/wp-content/uploads/pdf/pdf-revista-vol3/vol3-n1-portada.pdf” target=”blank” style=”flat” background=”#F38546″ color=”#ffffff” size=”6″ radius=”0″ icon=”icon: arrow-down” icon_color=”#ffffff” text_shadow=”0px 0px 0px #ffffff”]REVISTA VOL-3[/su_button][/su_column] [/su_row]
The impact of maternal prenatal complications on verbal fluency measures in children with ADHD: an empirical study
Objective: Attention Deficit/Hyperactivity Disorder (ADHD) has been linked to prenatal conditions and history of maternal pregnancy problems. While research has also shown a trend of reduced semantic fluency performance in children with ADHD, pediatric fluency studies are limited. The present study sought to evaluate whether maternal pregnancy problems are associated with impaired performance on measures of verbal fluency, including phonetic, semantic and category switching in children diagnosed with ADHD.
Methods: Thirty-Four culturally diverse patients between 8 to 18 years of age received a comprehensive neuropsychological evaluation at the pediatric neuropsychology and rehabilitation department of a major public South Florida Hospital between 2009 and 2017. A MANCOVA was conducted with pregnancy problems as the factor, performance on verbal fluency measures as the dependent variable, and age, ethnicity, gender and education as covariates.
Results: In category fluency and category switching the without pregnancy complication group performed better when compared to the with pregnancy complications group. There were not significant differences between groups in letter verbal fluency.
Conclusions: The preliminary results were commensurate with current literature identifying semantic fluency and category switching as areas of verbal executive dysfunction in a pediatric population with ADHD. Moreover, results demonstrated clinically significant relevance for subjects in a diverse clinical sample with comorbid prenatal maternal conditions related to their performance on semantic fluency and category switching. Further research with a larger sample is recommended.
Key Words: verbal fluency, pregnancy problems, ADHD, executive control, verbal ability
Pregnancy Complications
There are approximately 6 million pregnancies each year in the United States1. While the U.S. pregnancy rate reached its lowest in 2009 (12% lower than in 1990), and the abortion rate was the lowest recorded between 1976 and 2009 (32% lower), there was an increase in the pregnancy rate in women over 30 years old2. A notable steady increase since 1991 has been noted on pregnancy rates of women aged 40-44 years old, with 18.1 out of 1000 in 20083, increasing the risk of pregnancy complications within this population. Pregnancy complications are one of the leading causes of developmental deficit in children in the United States1. While a direct link between specific maternal prenatal medical conditions and developmental deficits is difficult to establish given the cofounding elements that contribute to pregnancy complications in women, efforts to explore the relationship between prenatal maternal conditions and developmental deficits continue to gain national and international attention. The most common medical problems a woman may experience during pregnancy include anemia, urinary tract infections (UTI’s), hypertension, gestational diabetes, obesity and weight gain, infections, and hyperemesis gravidarum1. Relatedly, the most frequent pregnancy complications associated with early neonatal complications and risk of later neuropsychological problems include hypertensive disorders, diabetes, and infection4. The increase in maternal prenatal medical conditions and subsequent neurodevelopmental disorders in the offspring represent a current public health concern in the United States, particularly around women’s access to adequate prenatal care and early interventions.Consequently, researchers around the world have explored the impact of a wide range of pregnancy complications across different cognitive domains, including gestational diabetes and communication skills5, maternal concentrations of thyroid-stimulating hormone (THS) and mild and severe hypothyroxinemia and expressive vocabulary functioning6, obesity and developmental disabilities and difficulties with emotions and relationships with other children7, high concentrations of environmental pollutants, specifically on polychlorinated biphenyls (PCBs) and hexachlorobenzene (HCB) and perception and working memory, very low gestational age (VLGA) and visuospatial sensorimotor processing and attention-executive functions8 and fetal growth restriction (FGR) and language, memory and learning 8, among others. In summary, pregnancy complications have shown to elevate risk of associated adverse medical, psychosocial, and behavioral outcomes in children.
Attention Deficit Hyperactivity Disorder
Neurodevelopmental disorders are disabilities associated primarily with the functioning of the neurological system and brain 9. Examples of neurodevelopmental disorders in children include ADHD, autism spectrum disorder (ASD), specific learning disabilities, and intellectual disability. The Center for Disease Control and Prevention CDC 1 estimates that 1 out of 6 children have one or more developmental disability or other developmental delay in the United States. As of 2016, 6.1 million children between 2 and 17 years living in the United States have been diagnosed with ADHD10, while 64% of children with ADHD also had a comorbid mental, emotional, or behavioral disorder 10.
ADHD is a complex, brain-based, neurodevelopmental disorder likely developing from the interaction of several social, developmental, environmental, and genetic risk factors11. Scientists study cause and risk factors in an effort to find better ways to manage and reduce the chances of a person having ADHD. Although ADHD is a multiple pathophysiological entity with a rather complex etiology, evidence supporting its biological foundations is the strongest among all psychiatric disorders11. While not causative, psychosocial hardship seems to also exacerbate the ADHD symptom severity and clinical presentation12. A large body of evidence supports a causal model in which numerous genetic and environmental factors network during early development to create a neurobiological susceptibility to the disorder13. Similarly, the genetic basis of ADHD proposes that gene regulating neurotransmitters systems have been implicated in the development of ADHD. While it is widely acceptable that genetic factors are involved in ADHD, there is still much to understand about the mechanism of action 14.
The pathogenesis of ADHD has also been associated to prenatal environmental factors 14. For example, maternal smoking has been found to produce a 2.7 fold increased risk for ADHD15. Nicotine receptors are known to modulate dopaminergic activity, which is believed to be involved in the pathophysiology of ADHD16. Similarly, children exposed prenatally to alcohol can become hyperactive, disruptive, and impulsive17; which is founded on the premise that prenatal alcohol exposure induces brain structural anomalies, especially in the cerebellum18. Other prenatal factors associated with ADHD speak to the role of malnutrition and dietary deficiency. Specifically, it has been suggested that an imbalance of essential fatty acid (omega-3 and omega-6) intake is involved in attention dysregulation19.
A Closer Look at ADHD and Pregnancy Problems Over Time
Several studies have explored the relationship between complications during pregnancy and ADHD. For instance, Rodriguez et al.,20 followed a group of 12,556 children between 7 and 12 years of age who were exposed to pre-pregnancy overweight and obesity, establishing an association to ADHD, which was diagnosed using the teacher rated Strength and Difficulty questionnaire (SDQ) and the Rutter Scale-second version (RB2). In 2010, Motlagh et al.21 concluded that individuals with ADHD alone had the highest rate of heavy maternal smoking (17.3%) and psychosocial stress (17.3) during pregnancy in a group of 222 between the ages of 7 and 18 diagnosed via the DuPaul-Barkley ADHD parent rating scale. Similarly, Marks et al.22 explored the emotional, cognitive, and intellectual functioning of a group of 212 children between 3 and 6 years of age whose mothers’ experienced gestational diabetes. Findings of this study suggested significantly worse language, visuospatial, and verbal skills as well as less developed full scale intellectual quotient (FSIQ), inhibition, and impulsivity at 3-4 years. At 6 years, however, gestational diabetes was associated with increased risk for ADHD based on the Conners’ parent rating scale in children of low socio-economic status.
In 2015, Modesto et al.23 looked at hypothyroxinemia during pregnancy in a group of 127 8-year olds, where hypothyroxinemia in early pregnancy was associated with higher scores (7%) for ADHD symptoms using the Conner’s parent rating scale-revised after adjustments for child and maternal factors. In 2016, Werenberg-Dreier et al. identified an increase in ADHD risk diagnosed via registry information following fever during gestational weeks 9-12 and genitourinary infections in weeks 33-36 when following a group of 89,146 children between 4 and 10 years of age who were part of the Danish National Birth Cohort Study.
Man et al.24 identified a higher risk of ADHD via maternal report in a group of 5659 children between 6 and 14 years of age whose mothers had a history psychiatric disorders during pregnancy, even if the mothers never used antidepressants. Pohlabeln et al.25, as part of the identification and prevention of dietary and lifestyle-induced health effects in children and infants (IDEFICS) study, explored pregnancy complications in the mothers of a group of 15,577 children ages 2 to 11.9 with a supplementary medical questionnaire, and found that children whose mothers suffered from pregnancy induced hypertension had approximately a two-fold risk of developing ADHD.
Executive Functioning
Executive Function refers to higher order cognitive processes (e.g., organization, planning, set-shifting, inhibition, and working memory) believed to regulate goal-directed thoughts and actions26. These same functions are needed for problem solving and maintaining goals27.
Executive functions have been historically associated with cortical networks involving frontal lobe functioning28. More specifically, an important neurological link has been established between the prefrontal cortex, executive functions and inhibition29. Other brain areas believed to impact executive function are the striate nodes (caudate nucleus), the anterior cingulated cortex, and the cerebellum29. Based on the neurobiological basis of ADHD, children with deficit in attention and/or hyperactivity disorder are expected to present with significant executive dysfunction. The first theoretical approach of atypical executive function in ADHD was Barkley’s30 model of inhibition dysfunction with subsequent disruption of other executive components. There is a general understanding that executive functions involve three basic components: (a) inhibition, which includes self-control; b) working memory; and c) cognitive flexibility or shifting31,32. Executive profiles of ADHD have been found to have large heterogeneity33, which makes executive function components unique and diverse. Furthermore, deficits in inhibitory control are very frequently reported in ADHD34,35,32. Other ADHD studies have found dysfunction in working memory; however, this particular dysfunction is observed with caution given that working memory deficits are also found in many other developmental disorders36. Interestingly, children with ADHD, even without co-occurring learning disorders, have also demonstrated impairment in visual-spatial storage and in verbal and visual-spatial central executive processes; however, intact verbal function is maintained35,37.
Verbal Fluency and ADHD
The performance of children with ADHD on measures of verbal fluency has been explored for decades. Multiple studies have been conducted across different age groups and using different verbal fluency measures yielding diverse performance results. For instance, Koziol et al.38 looked at the performance of 19 children between 7 and 14 years on the Knight Verbal Fluency measure and found that ADHD children performed significantly lower than expected, and lower than non-ADHD. In 1999, Cohen et al.39 explored the performance of 23 children ages 6 to 12 on the C.B.P.R verbal fluency test and found the performance of children with ADHD to be generally intact. Mahone et al.40 assessed 46 children aged 6 to 16 using the verbal fluency subtest from the Neurodevelopmental Psychological Assessment – First Edition (NEPSY-I), with the ADHD group demonstrating weaker performance on both letter fluency and semantic word fluency. Similar results were obtained by Hurks et al.41 while exploring the performance of 20 children with an average age of 14.6 years on the NEPSY-I initial letter fluency (ILF) and semantic category fluency (SCF) subtests. Results indicated substandard performance in the ADHD group in the first 15 seconds of the ILF task. Children’s deficits in the ILF tasks were believed to be grounded on the premise that performance on ILF tasks is less automated than on the SCF task41.
In 2013, Abreu et al.42 found the performance of 44, 12-year old children with ADHD significantly poorer on the FAS task, particularly on the letter “F”. Also, in 2013, Takács et al.43 evaluated the performance of 22 children with ADHD between the ages of 8 and 12 using the NEPSY-I and identified deficits in semantic fluency and category switching. As observed, the existing literature has consistently identified verbal fluency deficits among ADHD populations, with variable ability levels detected between letter fluency, semantic fluency, and category switching.
Verbal fluency tasks have been commonly used in neuropsychological assessment to detect executive dysfunctions and lexical access44,33,45. Although many similarities are found between the letter fluency and category fluency tasks, subtle yet salient differences are also identified in terms of task difficulty. For instance, the category fluency task resembles everyday activity tasks (i.e., making a shopping list, putting things in order, following a recipe) where individuals can use existing connections between related concepts to access responses46. Because priming studies propose that language is represented semantically48, category fluency tasks are believed to be more representative of language processes. For instance, Sauzéon et al.49 found that participants with smaller vocabulary produced fewer words in the category fluency task, whereas Bragrad et al.50 found children with specific learning disorders (SLD), especially in reading, to have deficits with word finding difficulty, suggesting that deficits in language development are more strongly reflected in the performance on category fluency tasks.
In contrast, participants must retrieve words from a phonemic category in the letter fluency subtest, a task that requires suppression of semantic activation and development of more novel retrieval strategies51,52. These strategies are believed to be more closely associated with frontal lobe function. Moreover, damage in frontal lobe areas are associated with poor performance in both phonetic and semantic fluency tasks53. Given the association between pregnancy complications, ADHD and deficits in executive control, the purpose of this study is to explore the impact of pregnancy complications on the performance of verbal fluency measures in children with ADHD.
This empirical study retrospectively reviewed archival records of children between 8 and 18 years of age who received a neuropsychological evaluation at the pediatric neuropsychology and rehabilitation department of a South Florida Hospital between 2009 and 2017.
Participants
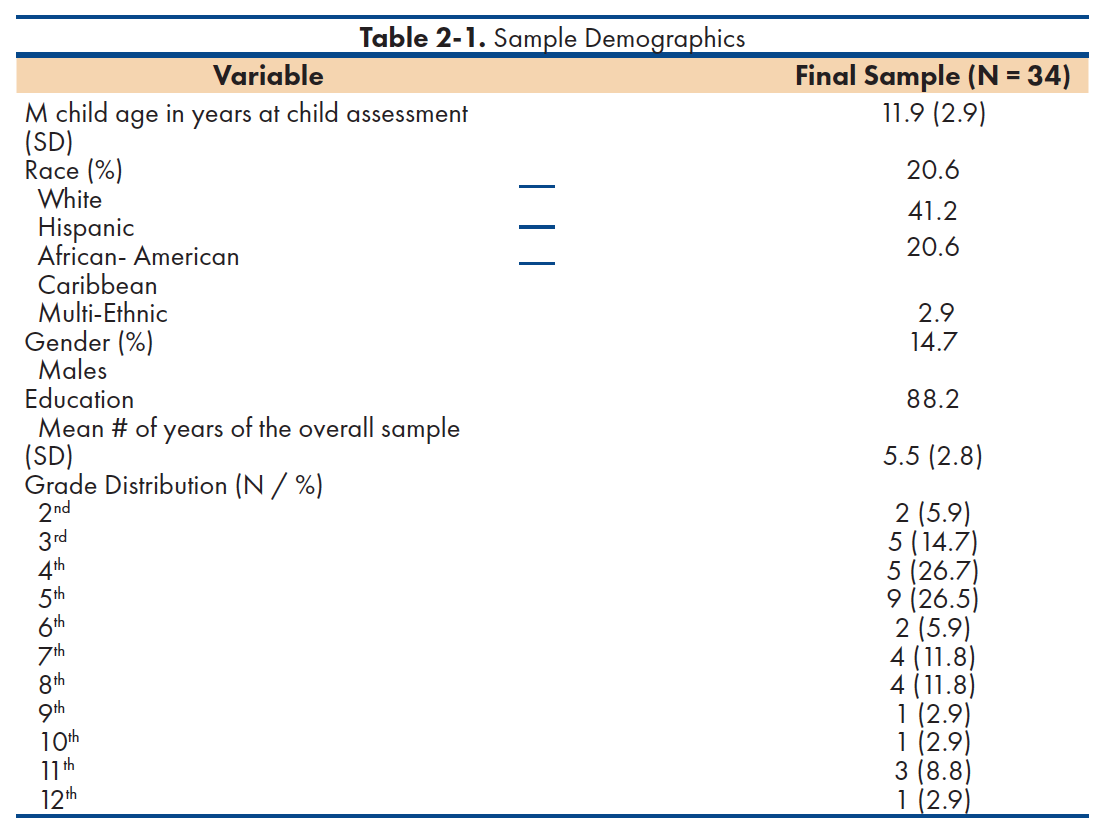
The participants in this study included 34 children selected on the basis of their ADHD diagnosis of which 88.2% were males (n=30), and 11.8% females (n=4), with a mean age of 11.9 (and a SD= 2.9) years. The average years of education for the overall sample was 5.5 (SD=2.8) years, with 26.5% of children reported to be in 9th grade (n=9). Race distribution included 20.6% White (n= 7); 41.2% Hispanic (n=14); 20.6% African – American (n=7); 2.9% Caribbean (n=1); and 14.7% multi-ethnic (n=5). All participants in this study were selected based on the following inclusion criteria: (a) have an ADHD diagnosis; (b) must have been diagnosed through a neuropsychological evaluation, and (c) have completed the letter fluency, category fluency and category switching tasks of the D-KEFS Verbal Fluency Subtest. Participants with history of a language disorder, co-morbid neurodevelopmental disorder or brain injury, and children whose mothers reported history of substance use or psychiatric conditions during pregnancy were excluded from the sample. The demographic information of the total sample can be found in detail on table 2-1.
Procedures
The sample group of the present study was collected from an ongoing parent study reviewing neuropsychological profiles of neurodevelopmental conditions in pediatric populations from a local hospital in South Florida, United States. Participants were referred internally or by local area pediatricians, pediatric psychiatrist, and local schools. Children received voluntary comprehensive neuropsychological evaluations in English. All participants and their parents signed a consent form that met Ethical and Institutional Review Board Standards. Participants were assured of the confidentiality of the data and that by no means they would be identified in the event of publication of research data. Completed de-identified evaluation results for all patients were entered into the neuropsychology and rehabilitation department’s database by trained research coders comprised of volunteers, practicum students, interns, and post-doctoral fellows.
Measures
Delis–Kaplan Executive Function System (D-KEFS)- Verbal Fluency Subtest
Children included in the study completed executive function tasks of verbal fluency including letter fluency, category fluency, and category switching tasks of the Delis-Kaplan Executive Function System (DKEFS). The D-KEFS comprehensively assesses higher level cognitive functions in both children and adults between 8 and 89 years. It measures executive functions such as attention, language and perception through 9 subtests. The Verbal Fluency subtest measures verbal knowledge, systematic retrieval of lexical items, monitoring, cognitive switching (flexibility), and establishing and maintaining cognitive set. It is comprised of letter fluency, which requires the patient to generate words by first letter; category fluency, where the patient is instructed to generate words by category; and category switching, which asks the patient to generate words alternating between two categories54. Scoring of the Verbal Fluency Subtest tasks was conducted following the D-KEFS manual.
Data Analysis
The total sample was divided into 2 groups based on the presence of maternal prenatal medical complications, resulting in a with pregnancy complication group and without pregnancy complication group. History of pregnancy complications was obtained via self-report or medical records at the time of the initial intake assessment through an unstructured interview. A comparative analysis using a one-way MANCOVA was conducted with pregnancy complications as the independent variable, verbal fluency tasks as the dependent variable, and ethnicity, age, gender, and education as the covariate variables. Regression of the covariate variables on the dependent variables aimed to eliminate the influence of covariates from analysis. A MANOVA on residual variance was then conducted to evaluate whether pregnancy complications influenced performance on verbal fluency tasks. A test of between subject effect was conducted to identify specific variance.
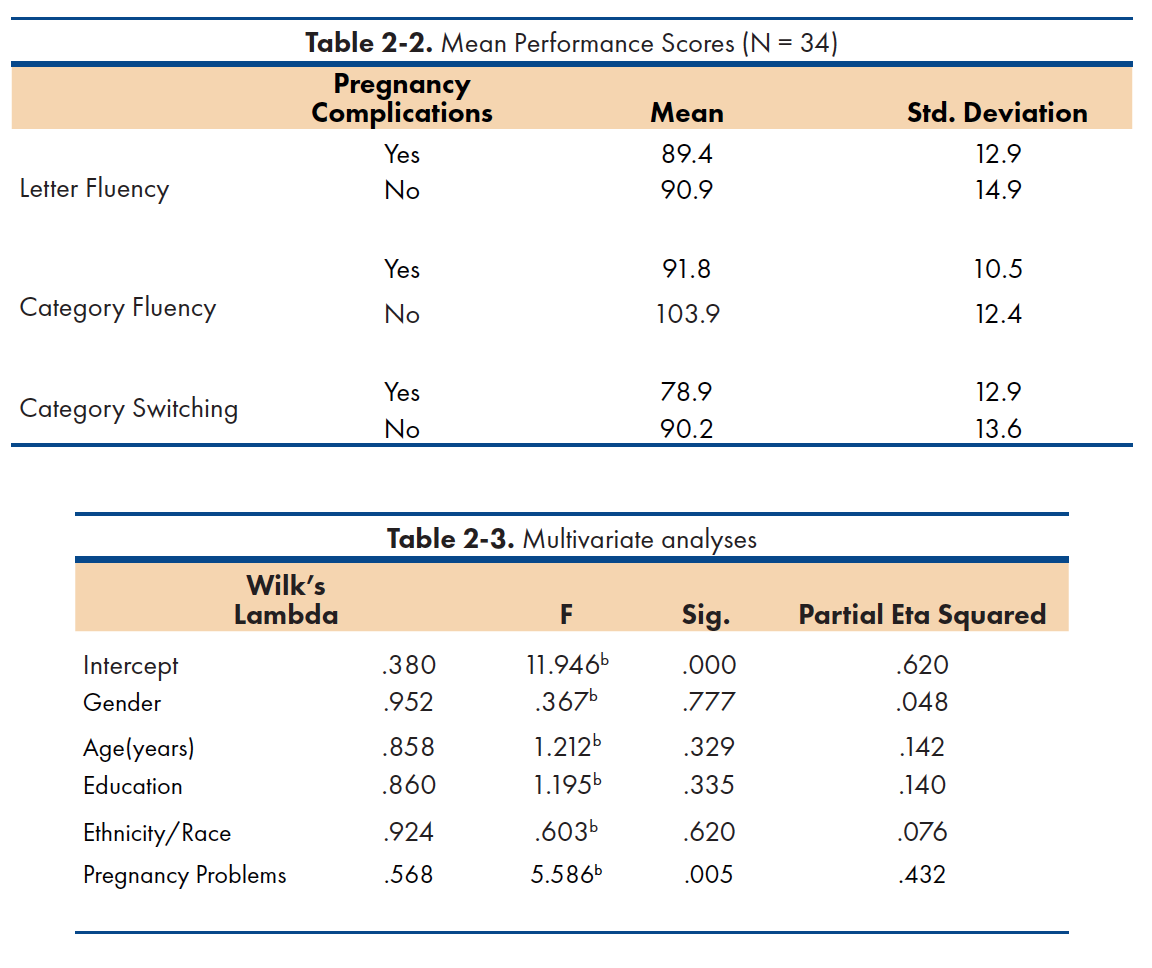
Mean versus performance descriptive statistics are reported on table 2-2. Between subject factors statistics identified 13 children in the with pregnancy complications group versus 21 children in the without pregnancy complication group. Broadly, children in the without pregnancy complications group demonstrated better performance across all tasks. As a group, the without pregnancy complications group demonstrated better performance on letter fluency (M=90.95; SD=14.88) when compared to the with pregnancy complications group (M= 89.44; SD=12.85). In category fluency, the without pregnancy complication group performed better (M=103.89; SD=12.44) when compared to the with pregnancy complications group (M=91.67; SD=10.52). Similarly, in category switching, children in the without pregnancy complications group performed better as a group (M= 90.24; SD=13.64) when compared to the with pregnancy complications group (M=78.89; SD=12.93). Notably, children in the with pregnancy complications group demonstrated better performance in the category fluency task, while their skills in the letter fluency and category switching tasks were evenly developed.
Multivariate test results are reported on table 3. A statistically significant difference was observed between groups on the combined dependent variables after controlling for gender, age, education, and ethnicity, F (3, 22) = 5.586, p < .05; Wilks' Λ = .568; partial η2 = .432.
Test of between-subject effect results demonstrate statistically significant differences between category fluency (p =.019), and category switching (p= .032) performance among groups, with a medium effect size (R2=.208 and R2=.177, respectively following partial eta squared values suggested by Richardson et al.55; and Cohen56 . In contrast, no statistically significant difference was observed in letter fluency performance between groups (p = .658).
Summary of Findings
This study aimed to explore the relationship between pregnancy complications and performance on measures of verbal fluency in children with ADHD. Out of a larger sample of 199 children, 34 children met inclusion criteria. The majority of children were males, Hispanic, school-aged, and in 5th grade (table 2-1). Three separate tasks of verbal fluency were administered in order to determine whether verbal fluency may have been particularly impacted by maternal pregnancy complications, and to determine performance variance. The with and without pregnancy complication groups had comparable demographic characteristics in terms of gender, age, ethnicity and years of education, which were not shown to mediate the effect of pregnancy complications on children’s performance among the three tasks.
To the best of our knowledge, this study is the first retrospective study exploring the relationship between ADHD and verbal fluency with pregnancy complications as an independent variable. As such, our study results are not entirely comparable with current literature without considering the impact of pregnancy complication as an additional performance variable. Our findings support the notion that pregnancy complications impact the performance of children with ADHD on measures of verbal fluency. A significant difference was found between groups in semantic fluency, which is comparable to performance patterns identified by Mahone et al.40 and Takács et al.43. Deficits in category fluency were also found to be similar to the findings of Takács et al.43, where significant difference in performance for the category switching task was observed. There could be a number of reasons why no significant performance difference was observed on letter fluency. It is possible that semantic skill development is more significantly impacted by developmental alterations during gestation. The absence of significant performance difference between groups on the letter fluency task can be explained by variability in the level of impact of pregnancy complications during gestation as well as difference in timing during gestation when mothers experienced complications. There is also the possibility that this particular verbal fluency task may be more sensitive to age, resulting in better performance over time. Given that close to 50% of our sample identified as Hispanic, it is possible that bilingualism and differences in language skills development may have played an important role in this ethnic group’s performance on semantic fluency tasks.
Implications
Given the established association between pregnancy complications and neurodevelopmental deficits, significant differences were expected between groups. In terms of overall performance, our findings were commensurate to the current literature where the without pregnancy complication group performance was consistently better than the with pregnancy complication group. However, as mentioned before, none of the existing literature included pregnancy complications as an independent variable, thus limiting our ability to compare our results to previous findings. Our results found common trends with Cohen et al.39 in that, although performance differences between both groups were identified, verbal fluency skills were not found to be impaired. With the exception of category switching, where the pregnancy complication group mean performance was very low, performance across groups ranged between low average to average.
In terms of ADHD performance patterns, the with pregnancy complication groups suggested clinically relevant implications for letter fluency (low average) and category switching (very low). That being said, our findings showed variability when compared to the present literature between ADHD and verbal fluency performance. For example, our outcomes differ from those observed by McGee et al.57 who did not identify an association between verbal fluency deficits and attention deficit disorder (ADD). Differences between outcomes can be explained by heterogeneity in sample population, where this research group was made up of only male participants, whereas our group, although mainly comprised by males, also included 20% females. Additionally, the age range for this study only included boys of 13 years of age, whereas our study had a range between 8 and 18 years of age. Interestingly, and different from current literature, pregnancy complication did not influence the performance of children on letter fluency tasks, suggesting that pregnancy complications were not a predictor of letter fluency dysfunction for this group. This particular finding is of great significance given the neurodevelopmental association of letter fluency with executive control as a frontal lobe function, which is consistent with the etiology of ADHD. However, it is important to highlight that the average performance for both groups on the letter fluency task was relatively lower when compared to category fluency task, but better than the category switching task. Letter fluency performance findings differ from deficits identified by Mahone et al.40. This difference could be attributed to the use of a different verbal fluency measure (NEPSY-I) whereas this study utilized the DKEFS’s Verbal Fluency subtests.
The current study obtained different outcomes when compared to findings by Hurks et al.41, who identified less developed letter fluency ability, particularly in the first 15 seconds of the task in the ADHD group. Because verbal fluency performance was coded as a scale score in our database, individual performance on each letter was not tracked. Therefore, we are unable to compare performance results on a letter by letter basis. Our letter fluency findings were also different to those obtained by Abreu et al.42 in that our groups did not evidence less developed letter fluency ability. Nevertheless, we did not assess an in-between letter comparison to identify difference in performance between letters.
Our results were also diverse in the category fluency task and were different from Hurks et al.41, who found no interaction between SCF performance and ADHD when compared to controls. Differences in results can be attributed to the absence of pregnancy complication as a variable from the analysis between groups. In contrast to Mahone et al.40, who found weaker verbal fluency performance in an ADHD sample, our results did not indicate less developed semantic fluency ability in any of the two groups when compared to the letter fluency and category switching tasks. In fact, both groups evidenced stronger performance in the category fluency task when compared to the other verbal fluency tasks. Our outcomes are also partially different from those found by Takács et al.43 who identified lower semantic performance on an ADHD sample, whereas we did not. These differences in results can be better explained by variability in their sample’s age range (8 to 12 years old); while our sample ranged from 8 to 18, suggesting that performance in this task may improve over time for this population. Our results were similar to McGee et al.57 who identified no deficits in verbal fluency in children with ADHD. Cross-sectional studies indicate that word fluency improves with increasing age to at least 13 years, with written word fluency increasing up to 18 years58. It is possible that the response organization and processing efficiency of children in the study who were younger than 9 years of age, the age at which frontal lobe changes related to executive function are believed to emerge, had not fully developed at the time the assessment was completed. The frontal lobe (FL) dysfunction model has been used to conceptualized ADHD given that the performance of children with ADHD differed significantly from normal controls on measures of frontal lobe function59. However, our results did not support this theory, and yielded opposite results to Moscovitch’s 60 hypothesis that patients with frontal lobe lesions are impaired on tests of letter but not category fluency.
In summary, our results evidenced important similarities as well as differences to findings in the current literature, specifically in the category fluency and category switching tasks. This variability in outcome is mainly attributed to differences in verbal fluency measures utilized to assess verbal fluency performance in previous studies as well as differences in age and gender among sample groups.
Limitations
The most important limitation of the present study is its retrospective nature. Having limited control over adherence to standardized test administration, proper documentation of maternal pregnancy complications, participants’ bilingual skills, and language functioning poses as significant drawback to maintaining uniformity among participants. Another limitation is the relative small size (N=34) of the ADHD sample meeting inclusion criteria. We believe the current study’s small sample size limits the generalization power of our results. Because the reporting of maternal pregnancy complications was conducted in a non-standardized fashion, there was significant variability in the way maternal pregnancy complication history was documented. This contributed to significant inconsistency among participants in terms of pregnancy complication history and specific medical conditions experienced by mothers during pregnancy. Participants were therefore unable to be grouped by medical condition or pregnancy complication type, and thus performance patterns within specific conditions could not be evaluated. The sample was not matched in terms of age and gender for the purpose of obtaining adequate sample size. As such, there is a rather large age-range variance between groups.
Although almost all of the average scores from the present sample are within a standard deviation from the standardization sample and clinically significant impairment was not observed, except in category switching, the study did not control for participants’ level of language functioning. Therefore, it is possible that variable language performance exists across both groups, thus impacting the results of the study, particularly in category switching. Given the diverse demographical characteristics of the sample and the geographic location where the assessment was conducted, it is no surprise that 40% of the sample was identified as Hispanic. Also a result of the socio-cultural characteristics of the geographic location is the expected bilingual nature of children’s language abilities. Because verbal fluency has also been identified as a function of verbal ability, language skills will play an important role in performance on these tasks. Although all children were assessed in English, following identification of language dominance through standard language proficiency testing, information on children’s bilingual skills and number of languages spoken at home was not available, and thus the extent by which bilingualism influenced performance on verbal fluency is unknown.
One of the biggest limitations involved inconsistencies found in the recording of maternal pregnancy history. For future research and clinical practice, the intake process should ensure consistency in how pregnancy history is documented, including specific questions added to existing and future parent history questionnaires to be filled out at initial contact. Future research on verbal fluency and pregnancy complications ought to be conducted within groups with specific pregnancy complications to identify patterns of performance within each pathology. Given our knowledge on language functioning and its identified relationship with category fluency performance, it will be crucial to control for language functioning to explore correlations and differences in performance in the presence of language dysfunction.
Conclusion
This study was founded on the understanding of atypical developmental expression as the result of changes experienced in utero that manifest throughout the lifespan. Much of what we know about pregnancy complications and neurodevelopmental deficits is grounded on neurobiological changes occurring at the cellular level during gestation. Because such changes occur at different times across the developmental spectrum, it is challenging to isolate with certainty which neurobiological mechanisms are responsible for atypical development in specific areas of functioning, resulting in a much more complex neuro-behavioral developmental assessment process. One of the important areas of human function that is believed to be impacted during gestation is executive control, guiding the focus of our study on defining the relationship between pregnancy complications and verbal fluency performance in a group of children with ADHD. The goal of this study was to identify the impact of maternal pregnancy problems on verbal fluency measures in children with ADHD. Age, gender, education, and ethnicity were not found to impact performance. A comparative analysis of covariance and subsequent test of between subject effect was conducted to identify specific variance was conducted. Broadly, children with ADHD without maternal history of pregnancy complications demonstrated better performance across all tasks. Our findings suggest a significant difference in performance on verbal fluency tasks when pregnancy complications are present, specifically on tasks of semantic fluency and set-shifting.
Although category fluency is one of the skills evidenced to be significantly different, this could be the result of the category fluency task having more resemblance to how language is stored in the brain, thus being more sensitive to language development variations during gestation. Because semantic fluency is associated with language function, future research must include language performance on ADHD cohorts to rule out other contributing factors impacting semantic fluency. Age, gender, education, and ethnicity did not seem to influence the effect of pregnancy complications on any of the verbal fluency tasks. Further investigation of verbal fluency performance within specific pregnancy complications sub-groups remains necessary
As observed in the existing literature, studies exploring the relationship between pregnancy complications and ADHD evidence clear differences in geographical location, timing, age ranges, and prenatal medical conditions. However, a consistent factor among all studies was the assessment method selected by researchers to assess for ADHD, which only included self-report measures from parents and teachers across all ages. Given the limitations previously discussed, future research should look at conducting similar studies with a larger sample size, as this will permit for results to be generalized to the rest of population.
- Centers for Disease Control and Prevention. Centers for Disease Control and Prevention; [cited 2019Oct18]. Available from: https://www.cdc.gov/reproductivehealth/maternalinfanthealth/pregcomplications.htm
- National Center for Health Statistics (2013). Pregnancy Rates for U.S. Women Continue To Drop. Retrieved on September 22, 2017 from https://www.cdc.gov/nchs/data/databriefs/db136.pdf
National Center for Health Statistics. Pregnancy Rates for U.S. Women Continue; [cited 2019Oct18]. Available from: https://www.cdc.gov/nchs/data/databriefs/db136.pdf
- National Vital Statistic Report (2012). Retrieved on September 22, 2017 from https://www.cdc.gov/nchs/data/nvsr/nvsr60/nvsr60_07.pdf
National Vital Statistic Report; [cited 2019Oct18]. Available from: https://www.cdc.gov/nchs/data/databriefs/db136.pdf
- Gerner G, Baron, IS. Pregnancy Complications and Neuropsychological
Outcomes: A Review. Child Neuropsychol. 2015;21(3):269-84.
- Dionne G, Boivin M, Séguin JR, Pérusse D, Tremblay RE. Gestational Diabetes hinders language development in offspring. 2008 Nov;122(5):e1073-9.
- Henrichs J, Bongers-Schokking JJ, Schenk JJ, Ghassabian A, Schmidt HG, Visser TJ, Hooijkaas H et al. Maternal thyroid function during early pregnancy and cognitive functioning in early childhood: the generation R study. J Clin Endocrinol Metab. 2010 Sep;95(9):4227-34.
- Jo H, Schieve LA, Sharma AJ, Hinkle SN, Li R, Lind JN. Maternal Pregnancy Body Mass Index and Child Psychosocial Development at 6 years of age. Pediatrics. 2015 May;135(5):e1198-209.
- Kallankari H, Kaukola T, Olsén P, Ojaniemi M, Hallman M. Very preterm birth and foetal growth restriction are associated with specific cognitive deficits in children attending mainstream school. Acta Paediatr. 2015 Jan;104(1):84-90.
- Basic Information about ACE [Internet]. EPA. Environmental Protection Agency; 2019 [cited 2019Oct18]. Available from: https://www.epa.gov/ace/basic-information-about-ace
- National Survey of Children’s Health – Data Resource Center for Child and Adolescent Health. [cited 2019Oct18]. Available from: https://www.childhealthdata.org/learn-about-the-nsch/NSCH
- McGough JJ. TDAH Oxford: Oxford University Press; 2014.
- Spencer TJ1, Biederman J, Mick E. Attention-deficit/hyperactivity disorder: diagnosis, lifespan, comorbidities, and neurobiology. Ambul Pediatr. 2007 Jan-Feb;7(1 Suppl):73-81.
- Sonuga-Barke EJ, Halperin JM. Developmental phenotypes and causal pathways in attention deficit/hyperactivity disorder: potential targets for early intervention? J Child Psychol Psychiatry. 2010 Apr;51(4):368-89.
- Emond V, Joyal C, Poissant H. Structural and functional neuroanatomy of attention-deficit hyperactivity disorder (ADHD). Encephale. 2009 Apr;35(2):107-14.
- Milberger S, Biederman J, Faraone SV, Chen L, Jones J. Is maternal smoking during pregnancy a risk factor for attention deficit hyperactivity disorder in children? Am J Psychiatry. 1996 Sep;153(9):1138-42.
- Potter AS, Newhouse PA, Bucci DJ. Central nicotinic cholinergic systems: a role in the cognitive dysfunction in attention-deficit/hyperactivity disorder? Behav Brain Res. 2006 Dec 15;175(2):201-11.
- D’Onofrio BM, Van Hulle CA, Waldman ID, Rodgers JL, Rathouz PJ, Lahey BB. Causal inferences regarding prenatal alcohol exposure and childhood problems. Arch Gen Psychiatry. 2007 Nov;64(11):1296-304.
- Coffin J, Baroody S,Schneider K, O’Neill J. Impaired cerebellar learning in children with prenatal alcohol exposure: a comparative study of eye blink conditioning in children with ADHD and dyslexia.Cortex. 2005; 41:389–398
- Raz R, Gabis L. Essential fatty acids and attention-deficit-hyperactivity disorder:a systematic review. Dev Med Child Neurol. 2009 Aug;51(8):580-92
- Rodriguez A, Miettunen J, Henriksen TB, Olsen J, Obel C, Taanila A et al. Maternal adiposity prior to pregnancy is associated with ADHD symptoms in offspring: evidence from three prospective pregnancy cohorts. Int J Obes (Lond). 2008 Mar;32(3):550-7
- Motlagh MG, Katsovich L, Thompson N, Lin H, Kim YS, Scahill L et al. Severe psychosocial stress and heavy cigarette smoking during pregnancy: an examination of the pre- and perinatal risk factors associated with ADHD and Tourette syndrome. Eur Child Adolesc Psychiatry. 2010 Oct;19(10):755-64
- Nomura Y, Marks DJ, Grossman B, Yoon M, Loudon H, Stone J et al. Exposure to gestational diabetes mellitus and low socioeconomic status: Effects on neurocognitive development and risk of attention deficit- hyperactivity disorder in offspring. Arch Pediatr Adolesc Med. 2012 Apr;166(4):337-43.
- Modesto T, Tiemeier H, Peeters RP, Jaddoe VW, Hofman A, Verhulst FC et al. Maternal mild thyroid hormone insufficiency in early pregnancy and attention- deficit/hyperactivity disorder symptoms in children. JAMA Pediatr. 2015 Sep;169(9):838-45.
- Man KKC, Chan EW, Ip P, Coghill D, Simonoff E, Chan PKL et al. Prenatal antidepressant use and risk of attention-deficit/hyperactivity disorder in offspring: population based cohort study. BMJ. 2017 May 31;357: j2350
- Pohlabeln H, Rach S, De Henauw S, Eiben G, Gwozdz W, Hadjigeorgiou C et al. Further evidence for the role of pregnancy-induced hypertension and other early life influences in the development of ADHD: results from the IDEFICS study. Eur Child Adolesc Psychiatry. 2017 Aug;26(8):957-967
- Zelazo P, Müller U. Executive function in typical and atypical development. The Wiley-Blackwell Handbook of Childhood Cognitive Development.2010
- Riba SSI, Fonseca-Pedrero E, Poch-Olivé ML. Early development of executive functions: A differential study. Anales de Psicología. 2015;31(2):607.
- Luria AR. The working brain: An introduction to neuropsychology (B. Haigh, trans). New York: Basic Books;1973.
- Lamm C, Zelazo PD, Lewis MD. Neural correlates of cognitive
control in childhood and adolescence: disentangling the contributions of age and executive function. Neuropsychologia. 2006;44(11):2139-48.
- Barkley RA. Behavioral inhibition, sustained attention, and executive functions: Constructing a unifying theory of ADHD. Psychol Bull. 1997 Jan;121(1):65-94.
- Diamond A. Executive functions. Annu Rev Psychol. 2013; 64:135-68.
- Miyake A1, Friedman NP, Emerson MJ, Witzki AH, Howerter A, Wager TD. The unity and diversity of executive functions and their contributions to complex “Frontal Lobe” tasks: A latent variable analysis. Cogn Psychol.2000 Aug;41(1):49-100.
- Sergeant JA, Geurts H, Oosterlaan J. How specific is a deficit of executive functioning for attention-deficit/hyperactivity disorder? Behav Brain Res. 2002 Mar 10;130(1-2):3-28.
- Castellanos FX, Sonuga-Barke EJ, Milham MP, Tannock R. Characterizing cognition in ADHD: Beyond executive dysfunction. Trends Cogn Sci. 2006 Mar;10(3):117-23.
- de Jong CG1, Van De Voorde S, Roeyers H, Raymaekers R, Oosterlaan J, Sergeant JA. How distinctive are ADHD and RD?: Results of a double dissociation study. J Abnorm Child Psychol. 2009 Oct;37(7):1007-17.
- Arnsten AF, Rubia K. Neurobiological circuits regulating attention, cognitive control,motivation, and emotion: disruptions in neurodevelopmental psychiatric disorders. J Am Acad Child Adolesc Psychiatry. 2012 Apr;51(4):356-67
- Martinussen R, Tannock R. Working memory impairments in children with attention-deficit hyperactivity disorder with and without comorbid language learning disorders. J Clin Exp Neuropsychol. 2006 Oct;28(7):1073-94.
- Koziol LF, Stout CE. Use of a Verbal Fluency Measure in Understanding and Evaluating Adhd as an Executive Function Disorder. Percept Mot Skills. 1992 Dec;75(3 Pt 2):1187-92.
- Cohen MJ, Morgan AM, Vaughn M, Riccio CA, Hall J. Verbal fluency in children: developmental issues and differential validity in distinguishing children with attention-deficit hyperactivity disorder and two subtypes of dyslexia. Arch Clin Neuropsychol. 1999 Jul;14(5):433-43.
- Mahone EM, Koth CW, Cutting L, Singer HS, Denckla MB. Executive function in fluency and recall measures among children with Tourette syndrome or ADHD. J Int Neuropsychol Soc. Enero de 2001;7(1):102-11.
- Hurks PP, Hendriksen JG, Vles JS, Kalff AC, Feron FJ, Kroes M et al. Verbal fluency over time as a measure of automatic and controlled processing in children with ADH. Brain Cogn.2004 Aug;55(3):535-44.
- Abreu N, Argollo N, Oliveira F, Cardoso A. L, Bueno JLO, Xavier, GF. Semantic and phonologic verbal fluency tests for adolescents with ADHD. Clin Neuropsychiatr: J Treat Eval. 2013;10(2):63-71.
- Takács Á, Kóbor A, Tárnok Z, Csépe V. Verbal fluency in children with ADHD: Strategy using and temporal properties. Child Neuropsychol. 2014;20(4):415-29.
- Matute E, Rosselli M, Ardila A, Morales G. Verbal and nonverbal fluency in Spanish-speaking children. Dev Neuropsychol. 2004;26(2):647-60.
- Tucha O, Mecklinger L, Laufkötter R, Kaunzinger I, Paul GM, Klein HE. Clustering and switching on verbal and figural fluency functions in adults with attention deficit hyperactivity disorder. Cogn Neuropsychiatry.2005 Jun;10(3):231-48.
- Shao Z, Janse E, Visser K, Meyer AS. What do verbal fluency tasks measure? Predictors of verbal fluency performance in older adults. Front Psychol.2014 Jul 22; 5:772.
- Collins AM, Loftus EF. A spreading-activation theory of semantic processing. Psychological Review. 1975;82(6):407–28.
- Jescheniak JD, Levelt WJM. Word frequency effects in speech production: Retrieval of syntactic information and of phonological form. Journal of Experimental Psychology: Learning, Memory, and Cognition. 1994;20(4):824–43.
- Sauzéon H, Raboutet C, Rodrigues J, Langevin S, Schelstraete A, Feyereisen P, et al. Verbal knowledge as a compensation determinant of adult age differences in verbal fluency tasks over time.J. Adult Dev. 2011;18:144–154.
- Bragard A, Schelstraete MA, Snyers P, James DG. Word-finding intervention for children with specific language impairment: a multiple single- case study. Lang Speech Hear Serv Sch. 2012 Apr;43(2):222-34.
- Luo L, Luk G, Bialystok E. Effect of language proficiency and executive control on verbal fluency performance in bilinguals. Cognition. 2010 Jan;114(1):29-41.
- Katzev M, Tüscher O, Hennig J, Weiller C, Kaller CP. Revisiting the functional specialization of left inferior frontal gyrus in phono-logical and semantic fluency: the crucial role of task demands and individual ability. J Neurosci. 2013 1 de mayo; 33(18):7837-45.
- Baldo JV, Shimamura AP. Letter and category fluency in patients with frontal Lobe lesions. Neuropsychology.1998 Apr;12(2):259-67.
- Delis DC, Kramer JH, Kaplan E, Holdnack J.Reliability and validity of the Delis- Kaplan Executive Function System: An Update. J Int Neuropsychol Soc. 2004 Mar; 10 (2):301-3.
- Richardson, J. Eta squared, and partial eta squared as measurements of effect size in educational research. Educational Research Review,2001;6:135-147.
- Cohen J. Statistical power analysis for the behavioural sciences. Academic; 1969.
- McGee R, Williams S, Moffitt T, Anderson J. A comparison of 13-year-old boys with attention deficit and/or reading disorder on neuropsychological measures. J Abnorm Child Psychol. 1989 Feb;17(1):37-53.
- Levin HS, Song J, Ewing-Cobbs L, Chapman SB, Mendelsohn D. Word fluency in relation to severity of closed head injury, associated frontal brain lesions, and age at injury in children. Neuropsychologia. 2001;39(2):122-31.
- Shue KL, Douglas VI. Attention deficit hyperactivity disorder and the frontal lobe syndrome. Brain Cogn. 1992 Sep;20(1):104-24.
- Moscovitch, M. Cognitive resources and dual task interference effects at retrieval in normal people: The role of the frontal lobes and medial temporal cortex. Neuropsychology.1994; 8:524–534.